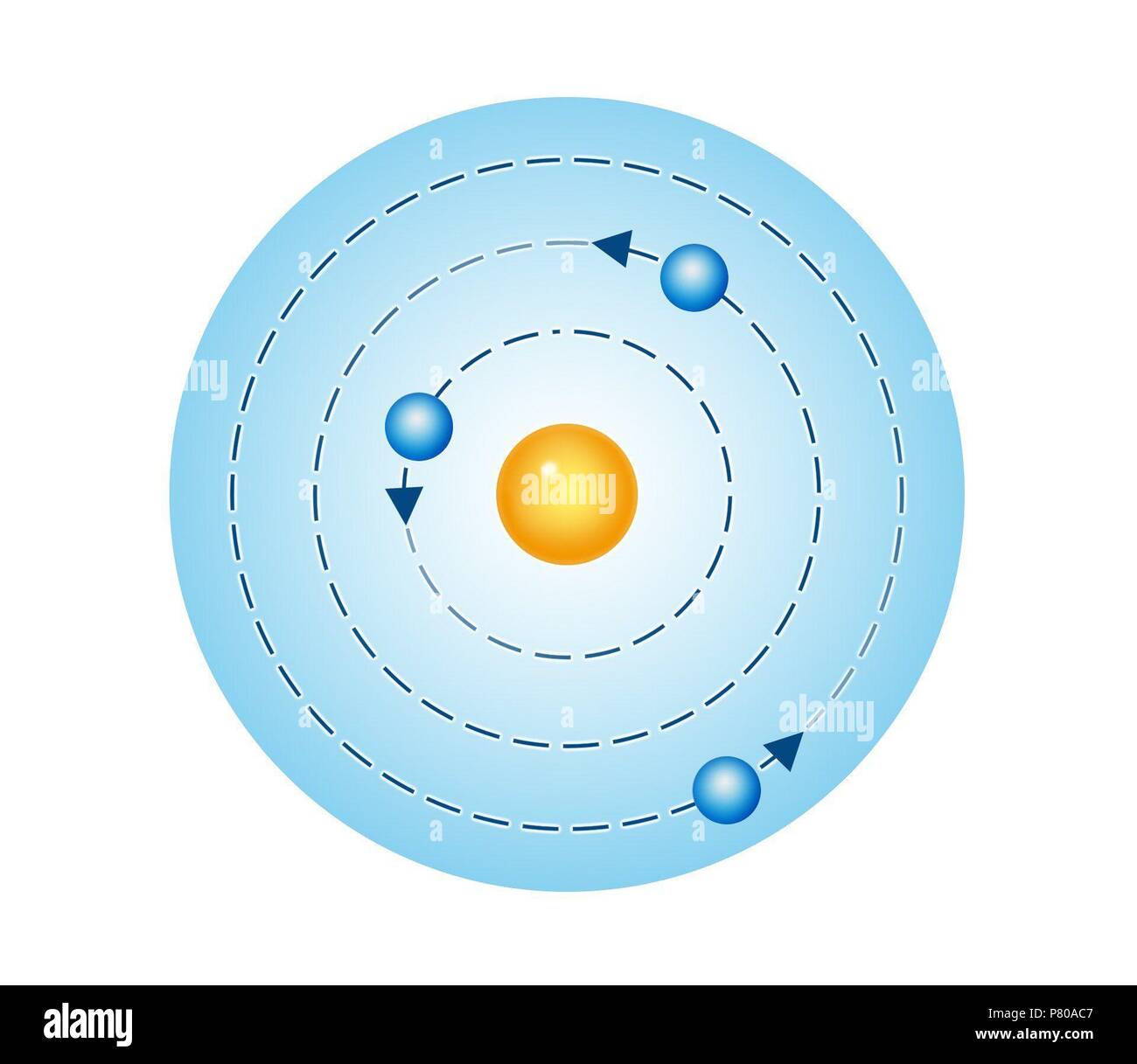
 Hence, energy becomes less negative as the electron is moved away. To move an electron further from the nucleus, energy must be imputed. 
Here q1 and q2 are charges of two different electronsĪn electron found in the ground state is assigned negative energy. The electric potential energy is given as: Electrons higher up are said to be in an excited state. The orbit closest to the nucleus has the lowest energy level and electrons in this orbit are said to be in the ground state. The energy that an electron has in an orbit is known as an energy level or energy state of an electron. We can calculate the maximum number of electrons in each orbit or shell in the same manner. Thus, the maximum number of electrons in the second orbit is 8.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
